Electrospinning has become a foundational technique in medical device materials engineering, enabling the fabrication of nanofiber mats with precisely tunable structural and functional properties. These materials are increasingly used in applications such as vascular grafts, wound dressings, drug-eluting coatings, filtration membranes, and tissue-contacting scaffolds, where clinical performance depends directly on consistent fiber morphology, mechanical integrity, and surface chemistry.
At the center of this process is the electrospinning system itself, which transforms polymer solutions into ultrafine fibers through the application of a high-voltage electric field. While this equipment is essential, successful electrospinning for medical devices requires far more than the spinning apparatus alone. Achieving reproducible, high-quality materials suitable for regulated environments depends on a deep understanding of solution behavior, process parameters, and final material characteristics. Without this foundation, even sophisticated electrospinning platforms remain vulnerable to variability that can compromise device performance, validation efforts, and regulatory confidence.
This is where supporting analytical instrumentation becomes indispensable. At Nanoscience Analytical, electrospinning is approached as an integrated, measurement-driven workflow rather than a single fabrication step. The richness of parameters makes electrospinning ideal for structured experimentation, including factorial design-of-experiments (DoE) approaches , where multiple variables are systematically varied to identify optimal processing windows and material performance targets.
By combining rigorous solution characterization, process optimization, and post-fabrication material analysis, we enable electrospinning to become predictable, repeatable, and scalable, qualities that are essential for medical device development and commercialization. This methodology reduces batch-to-batch variability, accelerates development timelines, and provides defensible data to support design controls, process validation, and quality assurance.
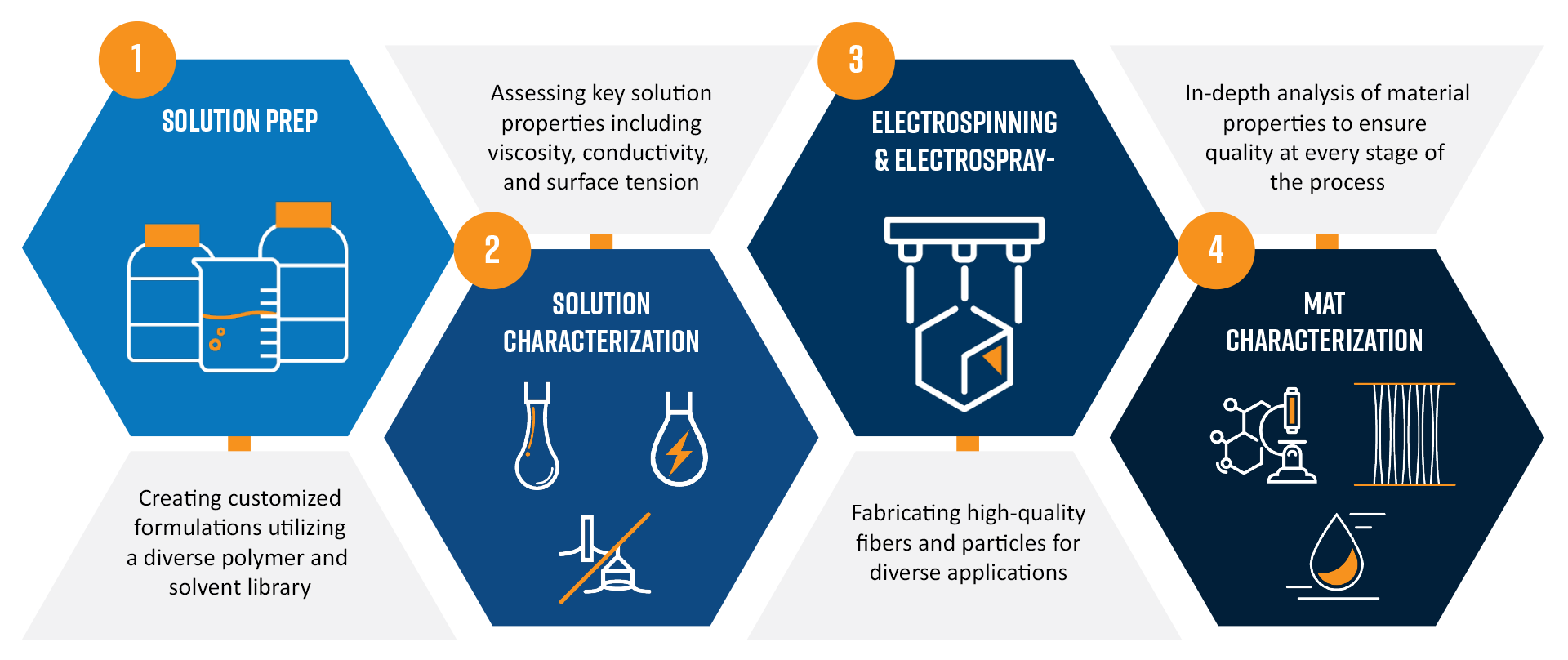
Solution Characterization: Building a Reliable Foundation for Regulated Applications
In medical device manufacturing, consistency is not optional. The properties of the polymer solution largely dictate jet stability, fiber formation, coating uniformity, and ultimately the safety and performance of the final device. Minor formulation inconsistencies can lead to significant changes in fiber diameter, porosity, drug loading behavior, or mechanical response, all of which can impact biocompatibility, release kinetics, or structural reliability.
For this reason, solution characterization serves as the foundation of reproducible electrospinning. Establishing defined solution specifications enables verification of formulation accuracy prior to processing, consistent performance across manufacturing lots, and early detection of off-spec material. This proactive control strategy minimizes costly rework, prevents downstream failures, and supports traceability and documentation expected in regulated medical environments.
Among the most influential solution parameters are surface tension, electrical conductivity, and viscosity. Each plays a distinct and interdependent role in fiber formation and process stability, and each must be measured and controlled with precision.
Surface tension governs the stability and elongation of the electrospinning jet. Excessively high surface tension resists deformation and stretching, promoting bead formation or discontinuous fibers, while surface tension that is too low can destabilize the jet and compromise coating uniformity on medical substrates. At Nanoscience Analytical, surface tension is measured using an optical tensiometer based on the inverted bubble method, a technique particularly well suited for polymer solutions that may be sensitive to evaporation or oxidation. By forming an air bubble within a sealed sample and analyzing its geometry using high-resolution imaging and software-based modeling, this method delivers precise, reproducible measurements while preserving solution integrity. Subtle variations in polymer concentration, solvent composition, or impurities that could affect coating consistency or device performance are readily detected.

Conductivity determines how effectively the applied electric field interacts with the solution and directly influences jet formation, stretching behavior, and fiber uniformity. In medical device applications, conductivity also affects the stability of coatings applied to complex geometries such as stents, guidewires, and implantable frames. Solutions with low conductivity often enable lower voltage operation and more controlled deposition, while excessively conductive systems may introduce instability and non-uniform coverage. Because conductivity is sensitive to polymer chemistry, solvents, additives, and temperature, it serves as both a development variable and a quality metric throughout scale-up and production.
Viscosity reflects the degree of polymer chain entanglement and is essential for producing continuous, defect-free fibers. Solutions that fall outside the optimal viscosity window can lead to beads, broken fibers, clogging, or irregular coating thickness, all of which complicate validation and compromise device reliability. By measuring viscosity under controlled shear and temperature conditions, formulation behavior can be standardized and controlled, ensuring that every batch entering production meets predefined performance criteria.
Mat and Coating Characterization: Verifying Structure, Function, and Integration
Once fibers are deposited, the next question is simple but critical: what was actually created?
Electrospun materials often exhibit complex morphologies, including variations in fiber diameter, alignment, surface texture, porosity, and coating thickness. These features strongly influence mechanical behavior, permeability, surface interactions, and functional performance, particularly for medical and biological applications.



Scanning Electron Microscopy (SEM) serves as a cornerstone of this evaluation. With nanometer-scale resolution and advanced image analysis capabilities, SEM enables precise visualization and quantitative assessment of electrospun materials. At Nanoscience Analytical, SEM is fully integrated into the electrospinning development workflow, allowing direct correlation between solution properties, processing conditions, and final device-relevant material outcomes.
This feedback is especially powerful in factorial studies, where multiple variables are adjusted simultaneously. SEM data makes it possible to identify which parameters drive meaningful changes and which have minimal impact, helping teams converge more rapidly on practical design spaces.
Through SEM-based analysis, polymer concentrations can be refined to eliminate defects, spinning parameters can be tuned to achieve targeted fiber diameters and coating thicknesses, and substrate coverage can be verified on complex device geometries. This continuous feedback loop transforms electrospinning from a trial-and-error technique into a controlled optimization process suitable for design verification and process validation.
Beyond basic morphology, we support more advanced characterization relevant to device-oriented applications. Cross-sectional imaging allows evaluation of coating thickness and layer-to-layer cohesion. High-resolution imaging can reveal how fibers interact with metallic or polymeric substrates, providing insight into adhesion behavior and coverage on complex geometries.
For composite or functional materials, elemental analysis and contrast techniques can be used to evaluate how nanoparticles, drugs, or fillers are distributed within the fibers or across the mat. Understanding whether additives are embedded, surface-localized, or heterogeneously dispersed is often essential when linking formulation choices to performance metrics.


By combining structural and compositional analysis, customers gain a clear picture of how experimental inputs translate into real material architectures.
Through SEM-based analysis, polymer concentrations can be refined to eliminate defects, spinning parameters can be tuned to achieve targeted fiber diameters and coating thicknesses, and substrate coverage can be verified on complex device geometries. This continuous feedback loop transforms electrospinning from a trial-and-error technique into a controlled optimization process suitable for design verification and process validation.
A Measurement-Driven Workflow for Medical Device Confidence and Control
Electrospinning offers exceptional flexibility for creating advanced medical device materials, but only when supported by rigorous measurement and characterization. From solution formulation to final coating validation, each stage requires precision, consistency, and defensible data. Analytical instrumentation is not optional in this environment; it is fundamental to quality systems, regulatory readiness, and patient safety.
By integrating solution characterization with comprehensive post-fabrication analysis, electrospinning becomes a predictable, data-driven methodology aligned with medical device development requirements. Measuring surface tension, conductivity, and viscosity establishes control before spinning begins, while SEM imaging and elemental analysis confirm that structural and compositional targets have been achieved after fabrication. Together, these capabilities support reproducibility across production lots, reduce development time, minimize material waste, and strengthen confidence in both device performance and regulatory submissions.
At Nanoscience Analytical, we provide the instrumentation, expertise, and integrated workflow needed to support electrospinning from early formulation through scale-up, validation, and quality assurance. Whether optimizing a drug-eluting coating, troubleshooting fiber defects on implantable devices, or preparing a process for manufacturing, our analytical approach enables reliable, high-performance results.
In medical device development, success is built measurement by measurement. With the right data, the right tools, and the right expertise, electrospun materials can move from promising laboratory concepts to safe, effective, and commercially viable clinical solutions.

